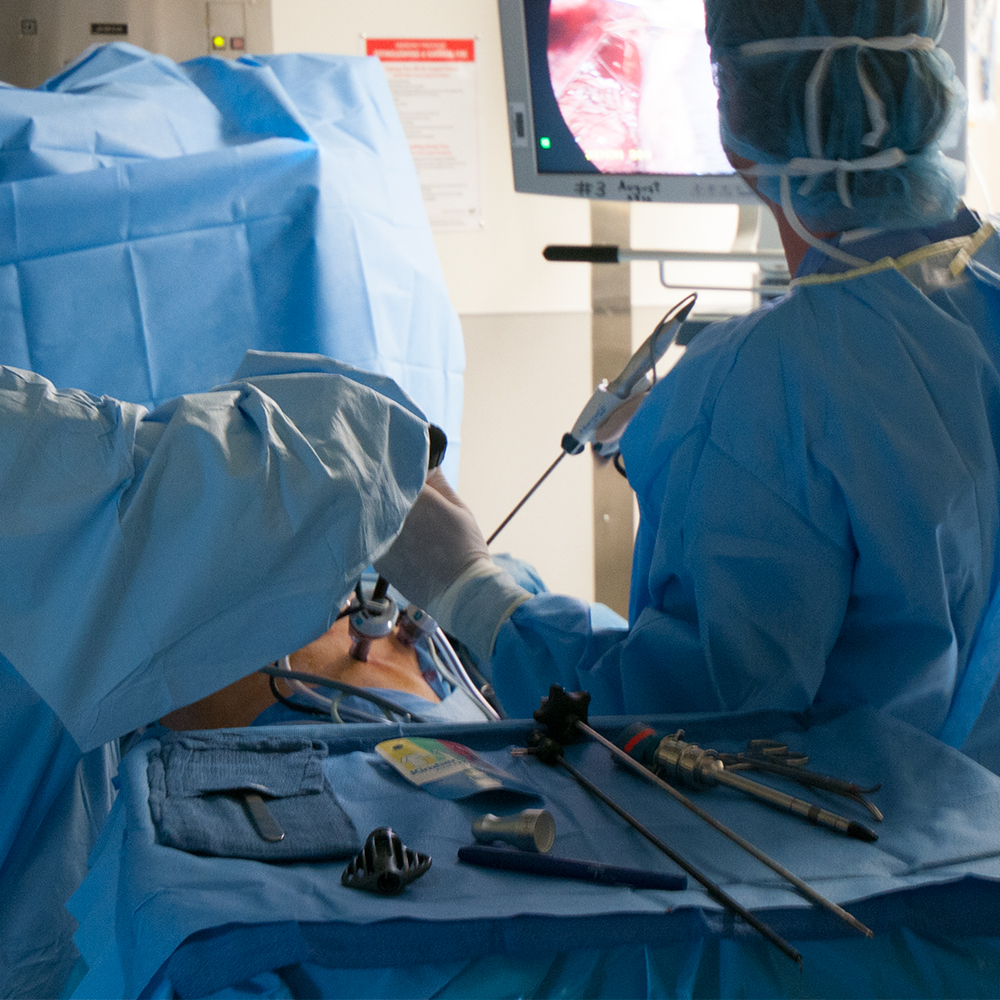
Laparoscopic Adrenalectomy
Adrenalectomy is the surgical removal of all or part of an adrenal gland. The most common indicator for adrenalectomy is the presence of hormone-producing adrenal tumors. Prognosis is dependent upon individual medical history.
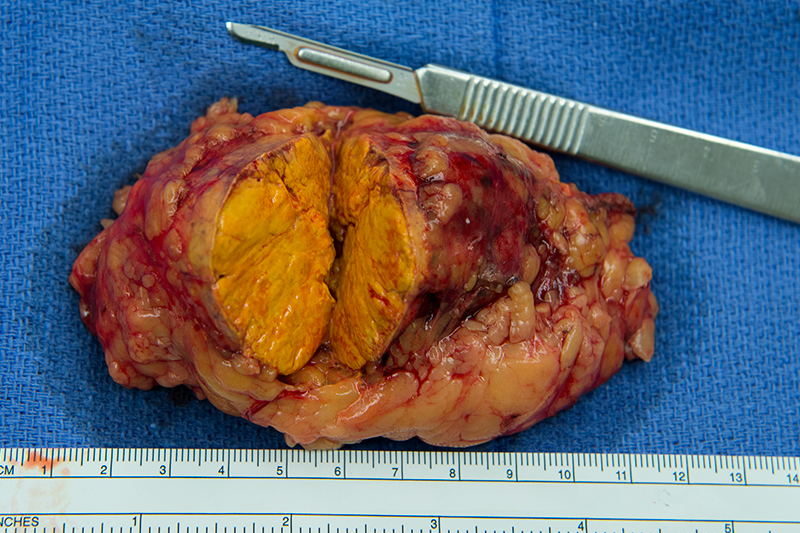
Golden tan color of an adrenal adenoma
What are the adrenal glands?
The adrenal or suparenal glands are a pair of glands that form part of the endocrine system, interacting with the hypothalamus and pituitary gland to regulate many processes within the body. They are located directly above the kidneys, within an area of the abdominal cavity known as the retroperitoneum. The right adrenal is roughly pyramidal and slightly smaller than the flatter and more crescent-shaped left adrenal. Each yellow-colored gland comprises two distinct parts: the cortex and the medulla.
What are the different parts of the adrenal cortex?
The adrenal cortex forms the outer portion of the gland. It secretes hormones including cortisol, aldosterone and androgens such as testosterone directly into the bloodstream. Cortisol controls the body’s use of proteins, fats and carbohydrates and also affects inflammation and other aspects of the immune response. Its production is regulated by adrenocorticotrophic hormone (ACTH), produced by the pituitary gland. Aldosterone controls blood volume and pressure by regulating absorption of electrolytes like sodium and potassium. Androgens are sex hormones. Present in males and females they are involved in development and maintenance of male characteristics, estrogen synthesis and regulation of processes in the kidney, liver, muscle, bone and reproductive organs.
What is the adrenal medulla?
The adrenal medulla is the inner part of the gland, responsible for production of the hormones norepinephrine (noradrenaline) and epinephrine (adrenaline), which affect heart rate, blood pressure, digestion and metabolism and are central to the fight-or-flight response to acute stress.
Diseases of the Adrenal Glands
Multiple conditions are associated with adrenal gland dysfunction, which may originate within the gland itself due to infection or disease, or externally, arising from disruption to the hypothalamic-pituitary-adrenal axis.
Among the most common reasons for adrenalectomy is the presence of adrenal nodules or adenomas. These benign growths or tumors are typically small and symptomless; observed as an incidental finding of imaging studies performed to investigate other issues. However, in some cases they can cause excess hormone production, leading to a variety of potentially serious health consequences. The symptoms caused by functioning adenomas are dependent upon the specific hormones produced.
Pheochromocytoma
Adrenal medulla tumors, or pheochromocytomas, cause excessive production of norepinephrine and epinephrine, frequently leading to hypertension. They are usually benign but can be cancerous. The presence of episodic headaches, heart palpitations and excessive sweating (diaphoresis or secondary hyperhidrosis) in a hypertensive individual is considered suggestive of pheochromocytoma, particularly if the individual has no other risk factors for hypertension. Other symptoms can include abdominal pain, anxiety, weight loss and nausea, although the tumors can be entirely symptomless.
Individuals with the inherited condition multiple endocrine neoplasia (MEN1 and MEN2) exhibit increased susceptibility to developing adrenal tumors. MEN 1 is associated with benign adrenal tumors, whilst MEN2 is associated with pheochromocytomas. Other genetic disorders associated with increased risk of developing pheochromocytomas include neurofibromatosis and Hippel-Lindau syndrome.
Aldosteronoma and Conn’s Syndrome
Overproduction of aldosterone or hyperaldosteronism causes sodium retention and excessive excretion of potassium in the urine. Primary hyperaldosteronism, sometimes termed Conn’s syndrome, can occur due to the presence of one or more aldosterone-producing tumors or aldosteronomas. Conn’s syndrome is typically associated with treatment-resistant hypertension and low blood potassium or hypokalemia is another common feature of the condition. Presenting symptoms can include excessive thirst, frequent urination, muscle weakness or cramps and visual disturbances.
Conn’s syndrome may also occur due to bilateral idiopathic adrenal hyperplasia (IAH); a condition in which both adrenal glands are enlarged, leading to increased hormone production. Hyperaldosteronism due to other causes, such as coronary or renal disease is termed secondary hyperaldosteronism. Treatment is dependent upon the underlying cause.
Cushing’s Syndrome
Cushing’s Syndrome, also known as hypercortisolism, is a collection of symptoms associated with prolonged exposure to elevated levels of cortisol. It can be iatrogenic; caused by long-term usage of high dosage corticosteroid medications or, less commonly, endogenous; occurring as a result of excessive cortisol production within the body. Many cases of endogenous Cushing’s syndrome arise due to the presence of an ACTH-producing tumor, either within in the pituitary gland, or located ectopically, in the lungs, pancreas or thyroid.
In some cases of endogenous Cushing’s syndrome the cause is an adrenal gland tumor, which is almost always benign. When adrenal cancer is involved, onset of symptoms is usually very rapid. Another cause of Cushing’s syndrome is primary pigmented micronodular adrenal disease (PPMAD). This inherited condition usually affects children and young adults, and is typified by the presence of multiple small cortisol-producing adrenal gland tumors.
Weight gain is the most common presenting symptom of Cushing’s syndrome. Other symptoms include central obesity, fatigue, muscle weakness, thin skin and excessive sweating (hyperhidrosis), and the condition is associated with depression and memory loss. Cortisol acts to alter body fat distribution so central obesity, a puffy face with enlarged chin and forehead known as “moon face”, and characteristic “buffalo hump” fat deposits on the back of the neck and shoulders may be observed.
Cushing’s syndrome shows a high degree of variability between individuals, with some experiencing rapid onset and severe symptoms whilst others display milder symptoms with a more gradual onset. Obesity, type 2 diabetes and hypertension are risk factors for developing Cushing’s syndrome.
Adrenal Cancer
Adrenal nodule carcinoma is a rare form of cancer. Tumors can develop in the cortex or the medulla and may be functioning or non-functioning. Tumor size is a strong indicator of malignancy, with tumors greater than approximately 2 inches (5cm) frequently found to be cancerous.
Most adrenal nodule carcinomas occur sporadically and can be difficult to diagnose. Functioning tumors may be detected due to the symptoms of hormone overproduction. Non-functioning tumors may only become apparent when they have grown large enough to cause physical sensations such as pain or abdominal fullness.
Adrenal neuroblastoma is an aggressive form of pediatric cancer, typically occurring before the age of two. Symptoms are often vague and it is common for metastasis to have occurred prior to diagnosis.
Most malignant growths within the adrenals occur due to the metastasis of breast, lung and other cancers, and are therefore not classified as adrenal cancer.
Hyperandrogenemia
Functional adrenal gland adenomas and carcinomas can cause overproduction of androgens, leading to symptoms such as irregular menses, growth of facial hair and loss of scalp hair in women and breast growth (gynecomastia) and loss of libido in men.
Adrenal Cysts
Adrenal cysts are hollow growths that may be filled with fluid, air or semisolid material. They are extremely rare and usually asymptomatic but, as with adrenal tumors, they can be functional and may cause abdominal pain and gastrointestinal symptoms by placing pressure on the abdominal organs. It is possible for adrenal cysts to rupture, causing internal hemorrhage and acute abdominal pain.
Indications for Adrenalectomy
Adrenalectomy may be total, involving complete removal of the gland, or subtotal, with only the affected area(s) removed. Preservation of cortical function is generally preferable, but may not be possible, depending upon the site and number of lesions present.
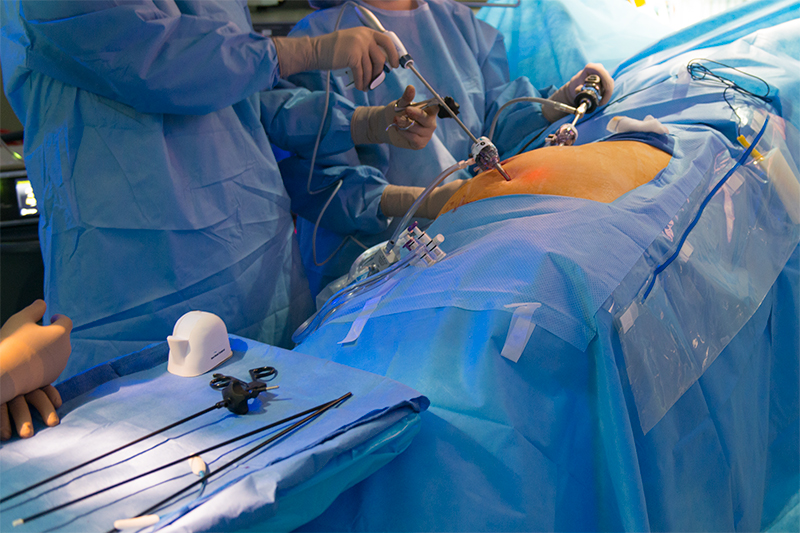
Port Placement and a Laparoscopic Adrenalectomy
In individuals with bilateral aldosteronomas, treatment with medication such as spironolactone or aldactone is often recommended as an alternative to surgery, in order to avoid complications arising from removal of both adrenals. Conversely, total bilateral adrenalectomy may sometimes be indicated in cases where both glands are healthy but the hormones they produce are aggravating another serious condition, such as breast or prostate cancer.
Adrenalectomy for Cushing’s syndrome is typically unilateral, although bilateral surgery may occasionally be indicated in the presence of high-risk symptoms such as poorly controlled hypertension.
Total adrenalectomy is usually indicated for malignant tumors, very large intra-adrenal tumors or cysts and cases where many lesions are present. Partial adrenalectomy may on occasion be considered safe and feasible for smaller tumors that are easy to differentiate from the normal gland.
For most incidentally detected non-functioning adrenal masses, monitoring rather than removal is preferred unless the lesion is greater than approximately 2 inches (4 to 5 cm).
Description of the Procedure
The adrenal glands are well suited to laparoscopic resection due to their small size and location within the body, enabling several possible surgical approaches. The most common approach is via the flank beneath the rib cage, requiring four incisions along the rib margin.
During the procedure multiple small incisions are made through which a laparoscope and various surgical instruments can be inserted. After the adrenal gland has been dissected it is placed within a sterile pouch. Slight enlargement of one of the incisions then facilitates removal of the pouch from the body.
Laparoscopic surgery is associated with reduced recovery times, shorter hospitalization, and improved cosmetic outcomes. It is therefore particularly recommended for cases of Cushing’s syndrome, which can cause poor wound healing.
Open surgery may be required if the position of the adrenals prevents adequate laparoscopic access, if a particularly large lesion is involved or when intracystic hemorrhage has occurred. Some surgeons may also favor an open approach for metastatic adrenal cancers.
The adrenal glands are highly vascular, so laparoscopic adrenalectomy may be contraindicated in the presence of blood clotting disorders. Other contraindications for a laparoscopic approach include morbid obesity and significant cardiovascular disease.
Postsurgical Considerations
Aftercare and duration of hospitalization will depend upon various factors including the type of procedure performed and the disease being treated.
Following laparoscopic adrenalectomy, hospital discharge may be possible the day after surgery. Recovery times following open surgery are typically longer and individuals with significant hypertension, such as those with pheochromocytomas, usually need to spend additional time under intensive observation.
Laparoscopic Incisions used for Adrenalectomy
When adrenalectomy is performed due to aldosteronoma, levels of aldosterone in the blood will be tested a day after surgery to confirm the procedure has been successful. Blood potassium levels will also be regularly checked for several weeks postoperatively and a high-sodium diet may be recommended in order to avoid hyperkalemia (excessive potassium), which can lead to serious cardiac complications. Blood pressure will be carefully monitored and hypertensive medications gradually tailed off.
Metanephrine is a catecholamine produced by breakdown of adrenaline. It is considered a biomarker for adrenaline producing tumors. Following surgery for pheochromocytoma, a blood or urine metanephrine test will be administered two weeks postoperatively, and at regular yearly intervals for up to a decade. As with aldosteronoma follow-up, blood pressure will also be monitored to facilitate appropriate gradual withdrawal of hypertensive medications.
Return to normal daily activities is usually possible within one to two weeks following laparoscopic surgery and four to six weeks after open adrenalectomy.
Adrenal cancers frequently warrant more intensive follow-up, possibly also requiring additional treatment with radiotherapy, radionuclide therapy or chemotherapy.
Prognosis after adrenalectomy
Prognosis following adrenalectomy for cancer is highly dependent on the stage of the disease and various other factors including age and general health. Outcomes are best for early-stage adrenal cancers but late diagnosis is common.
Adrenal surgery is curative for the majority of individuals with Cushing’s syndrome, Conn’s syndrome and non-malignant pheochromocytoma. However, the presence of a longstanding functional tumor can result in secondary effects such as damage to blood vessels, leaving residual heath issues. It is also possible for adrenal tumors to reoccur following subtotal adrenalectomy, although the risk is typically low unless a genetic predisposition exists. Lifetime follow-up is particularly recommended in such cases.
All individuals who have undergone total adrenalectomy, and many who have had part of their adrenals removed will need corticosteroid replacement therapy for life. The synthetic hormones fludrocortisone and hydrocortisone are commonly prescribed for this purpose.
In cases of unilateral disease, overproduction of hormones by the affected adrenal may have caused the contralateral gland to atrophy, and pituitary gland function may also have been suppressed. It is therefore common for corticosteroid supplementation to be recommended for up to a year following unilateral adrenalectomy, to allow recovery of the hypothalamic-pituitary-adrenal axis of the healthy gland.
Nelson’s Syndrome
Nelson’s syndrome is a tumor of the pituitary gland, occurring rapidly after bilateral total adrenalectomy in approximately five to ten percent of individuals. Symptoms include darkening of the skin, headaches and visual disturbances. Treatment involves surgical removal of the tumor, potentially followed by a course of radiotherapy.